Cerebral Palsy Might Be Detected in Toddlers with Glove by Exoskeleton Engineer
 Virginia Tech (VT) associate professor of mechanical engineering Pinhas Ben-Tzvi, will develop a robotic glove to facilitate the early diagnosis of cerebral palsy in children.
Virginia Tech (VT) associate professor of mechanical engineering Pinhas Ben-Tzvi, will develop a robotic glove to facilitate the early diagnosis of cerebral palsy in children.
 Ben-Tzvi, founding director of the VT Engineering department’s Robotics and Mechatronics Laboratory, worked for years on the development of robotic assistive exoskeletons, and on medical diagnostic robotics systems from 2000 to 2002 at General Electric Medical Systems.
Ben-Tzvi, founding director of the VT Engineering department’s Robotics and Mechatronics Laboratory, worked for years on the development of robotic assistive exoskeletons, and on medical diagnostic robotics systems from 2000 to 2002 at General Electric Medical Systems.
He took on his newest challenge  as proposed by Dr. Nathalie Maitre, principal investigator in the Maitre Lab at Nationwide Children’s Hospital in Columbus, Ohio. Maitre asked Ben-Tzvi to investigate whether his Sensing and Force-Feedback Exoskeleton (SAFE) robotic glove could be modified to assist in the early diagnosis of cerebral palsy in children.
as proposed by Dr. Nathalie Maitre, principal investigator in the Maitre Lab at Nationwide Children’s Hospital in Columbus, Ohio. Maitre asked Ben-Tzvi to investigate whether his Sensing and Force-Feedback Exoskeleton (SAFE) robotic glove could be modified to assist in the early diagnosis of cerebral palsy in children.
“Doctor Maitre proposed to fit a modified version of my glove to a child in an effort to apply somatosensory stimuli to measure cortical responses for the early detection of cerebral palsy,” Ben-Tzvi said in a VT press release.
The SAFE device for adult hands will be re-created and assessed first in a larger size before it is scaled down to fit the hand of a little child.
“There’s a couple of really challenging aspects to this project,” Ben-Tzvi said. “The first, of course, is the typical challenge we all face in making something work. But then we have two additional engineering challenges here to make it much smaller and to make it look child-friendly so it doesn’t scare the children who have to use it.”
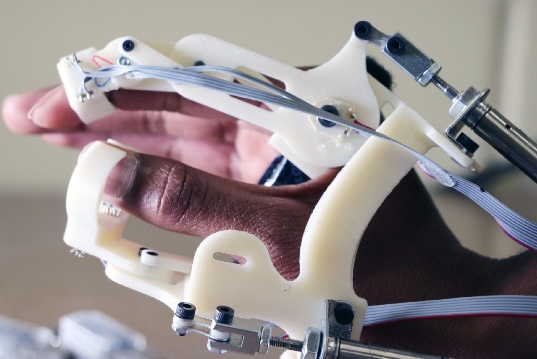
The robotic glove, partially produced with additive manufacturing technologies, features a pneumatically actuated index finger and thumb to allow for a pincer grip that can be measured as a response time. Photo courtesy: Virginia Tech
The SAFE glove currently features electrically or pneumatically actuated “fingers” that connect to a wrist-mounted base-plate and to the fingertips via straps. The device is actuated with miniature DC motors through antagonistically routed cables at each finger, which act as both active and passive force actuators. The glove is a wireless and self-contained mechatronic system that mounts over the dorsum of a bare hand, and provides haptic force feedback to each finger.
The development plan for a smaller version of the glove will be executed in two phases.
The first phase will fit the device on hands of children 12-36 months old using only a thumb and forefinger to initiate a pincer grasp. Because the design glove will provide tactile feedback, the child will be wired with brain sensors connected to the hospital’s Net Station software electroencephalogram (EEG) monitoring, while the robotic device initiates the pincer grasp and pools data that will include finger position and tactile information. The EEG recording of brain wave patterns will detect electrical activity abnormalities in the brain.
In phase two, toddlers will be tested in a study to establish baseline values for cortical and sensorimotor responses in 20 children who are developing normally, and five with diagnosed cerebral palsy.
“There is a good deal of potential for the device, and its an interesting application of a product that was mainly intended for hand rehabilitation for stroke victims,” Ben-Tzvi said. “I think anytime your work can be modified to help identify a medical issue more quickly, the more beneficial it becomes for the patient and the more options it allows for in treatment. The fact we are taking a device made to deal with after-the-fact issues and using it to possibly help make an earlier diagnosis of an issue is very exciting.”


